- 背景資料
葡聚醣(Glucan)為D-葡萄糖單體藉由糖苷鍵連接的多醣,依據糖苷鍵鍵結方式的差異,可分成α-葡聚醣和β-葡聚醣
常見的α-葡聚醣:
1️⃣直鏈澱粉(amylose)➡️α-1,4糖苷鍵
2️⃣支鏈澱粉(amylopectin)➡️α-1,4糖苷鍵+α-1,6糖苷鍵
3️⃣肝醣(glycogen)➡️α-1,4糖苷鍵+α-1,6糖苷鍵
常見的β-葡聚醣:
1️⃣纖維素(cellulose)➡️β-1,4糖苷鍵➡️植物的細胞壁
2️⃣卡德蘭膠(Curdlan)➡️β-1,3糖苷鍵➡️細菌性胞外多醣體
(1,3)(1,6)-β-D-glucan為真菌和酵母菌細胞壁的成分,在過去的科學研究當中,此種β-葡聚醣可能具有降低發炎反應、降低人體血清中低密度脂蛋白膽固醇含量、增加腸胃道功能或免疫調節作用
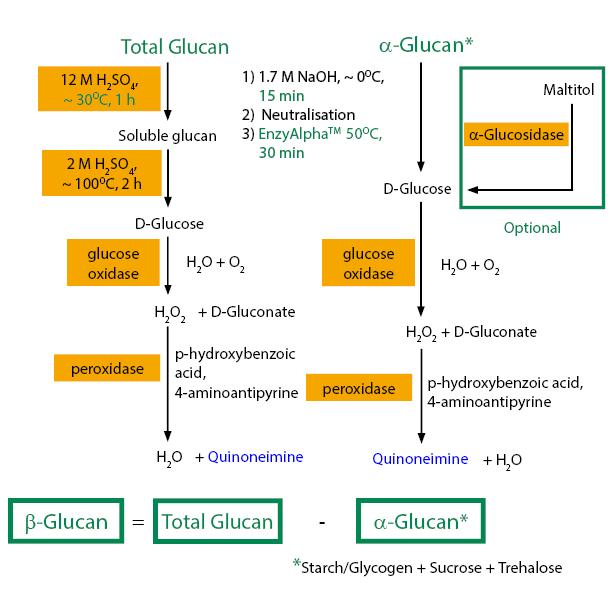
- A. MEASUREMENT OF TOTAL GLUCAN (α-glucan + β-glucan) 總葡聚醣測量值
The protocol below describes analysis of solid samples.
Liquid samples can be analysed by aliquoting 0.5 mL of sample in place of weighing the sample and proceeding with step 3. The calculation should be adjusted accordingly as described in the calculation section.
1. Mill mushroom or yeast sample to pass a 1.0 mm screen using a centrifugal mill.
2. Add the milled sample (approx. 90 mg, record exact weight) to a 20 x 12 mm culture tube. Tap the tube to ensure that all the sample falls to the bottom of the tube.
3. Add 2.0 mL of 12 M sulphuric acid to each tube, cap the tubes and stir vigorously on a vortex mixer. Place the tubes in a 30°C water bath and incubate for 1 h. Vigorously stir the tube contents for 10-15 sec every 15 minutes on a vortex mixer to ensure complete dissolution of the β-glucan.
4. Add 4 mL of distilled water to each tube, cap the tubes and vigorously stir the contents on a vortex mixer for 10 seconds. Then add 6 mL of distilled water, cap the tubes and stir the contents for a further 10 seconds.
5. Loosen the caps on the tubes and place them in a boiling water bath (~ 100°C). After 5 minutes, tighten the caps and continue the incubation for 2 hours. Vigorously stir the tube contents for 10-15 seconds every 30 minutes on a vortex mixer.
6. Cool the tubes to room temperature (~ 15 minutes) and carefully loosen the caps.
7. Quantitatively transfer the contents of each tube to a 100 mL volumetric flask using 200 mM sodium acetate buffer (pH 4.5) to rinse the tubes.
8. Add 6 mL of 8.0 M NaOH solution to the volumetric flask and adjust to volume with 200 mM sodium acetate buffer (pH 4.5). Mix the contents thoroughly by inversion.
9. Filter an aliquot of the solution using either filter paper (i.e. Whatman® filter paper Grade 201 or equivalent) or syringe filters (Corning® 0.2 µm or equivalent). Alternatively, centrifuge an aliquot of the solution at 15,000 g for 5 minutes.
10. Transfer 0.1 mL aliquots (in duplicate) of clarified extract to the bottom of glass test tubes (16 x 100 mm).
11. Add 3.0 mL of GOPOD Reagent to each tube and incubate at 40°C for 20 minutes.
12. Measure the absorbance of all solutions at 510 nm against the reagent blank.
Determination of Hydrolysis Correction Factor (HCF) - Optional:
The glucose Hydrolysis Correction Factor can be experimentally determined by comparing pure glucose (D-Glucose CAS:50-99-7; >99 % purity, not supplied) recovery levels before and after acidic treatment. The HCF must be applied to each
set of determinations to account for loss of glucose during the acidic hydrolysis procedure. Alternatively, a value of 1.05 been repeatably achieved and can be used in the calculations for Total Glucan - see associated validation report for more
information.
1. Weigh 90 mg of pure glucose into a 20 x 125 mm culture tube and run Method A as described above to generate a value for Glucose (% w/w) with acid.
2. In parallel, weigh 90 mg of pure glucose into another 20 x 125 mm culture tube and run the method described above replacing all volumes dispensed with distilled water to generate a value for Glucose (% w/w) without acid.
3. Hydrolysis Correction Factor (HCF) Calculation:

- B. MEASUREMENT OF α-GLUCAN(α-葡聚醣測量值)
The protocol below describe analysis of solid samples.
Liquid samples can be analysed by aliquoting 0.5 mL of sample in place of weighing the sample and proceeding with step 2. The calculation should be adjusted accordingly as described in the calculation section.
1. Add the milled sample (approx. 100 mg, record exact weight) to a 20 x 125 mm culture tube. Tap the tube to ensure that all the sample falls to the bottom of the tube.
2. Add 0.2 mL of 80% Ethanol (v/v) and stir the tubes in a vortex mixer.
3. Add a magnetic stirrer bar (5 x 15 mm) followed by 2 mL of ice cold 1.7 M NaOH to each tube and suspend the pellets by stirring for 15 minutes in an ice water bath over a magnetic stirrer.
4. Add 8 mL of 1.2 M sodium acetate buffer (pH 3.8) containing 5 mM Calcium Chloride to each tube with stirring. Immediately add 0.3 mL of Bottle 1 [EnzyAlpha], mix thoroughly and place the tubes in a water bath at 50°C.
5. Incubate the tubes at 50°C for 30 minutes with intermittent mixing on a vortex stirrer.
6. Remove the tubes from the incubator and let them equilibrate to room temperature (~ 15 minutes).
7. Centrifuge an aliquot of the solution at 15,000 g for 5 minutes. Alternatively, samples can be clarified using a filter paper (Whatman filter paper Grade 201 or equivalent) or a syringe filter (Corning® 0.2µm or equivalent).
8. For samples containing > 10 % α-glucan content; add 1 mL of the sample solution to 9 mL of distilled water, mix well and add 0.1 mL in duplicate to the bottom of glass test tubes (16 x 100 mm) for analysis using GOPOD Reagent.
NOTE: a Dilution factor (Df) of 10 should be included in the calculation.
9. For samples containing < 10% α-glucan content; directly transfer 0.1 mL of the sample solution in duplicate to the bottom of glass test tubes (16 x 100 mm) for analysis using GOPOD Reagent.
10. Add 3.0 mL of GOPOD Reagent to each tube and incubate at 40°C for 20 minutes.
11. Measure the absorbance of all solutions at 510 nm against the reagent blank.
商品特色
商品規格
- 商品規格(100個檢測反應)
Bottle 1: ( 2 x 15 mL )
EnzyAlpha™
Contains sodium azide (0.02% w/v) as a preservative.
Store below -10 °C. See individual label for expiry date.
Bottle 2:
GOPOD Reagent Buffer
Contains sodium azide (0.09% w/v) as a preservative.
Store at 4°C. See individual label for expiry date.
Bottle 3:
GOPOD Reagent Enzymes
Freeze-dried powder.
Store below -10°C. See individual label for expiry date.
Bottle 4: (5mL)
D-Glucose standard, (1.0 g/L)
Contains 0.2% (w/v) benzoic acid as a preservative.
Store sealed at room temperature. See individual label for expiry date.
Bottle 5: (2g)
Yeast Glucan Control
Glucan content stated on the bottle label.
Store at 4°C. See individual label for expiry date.