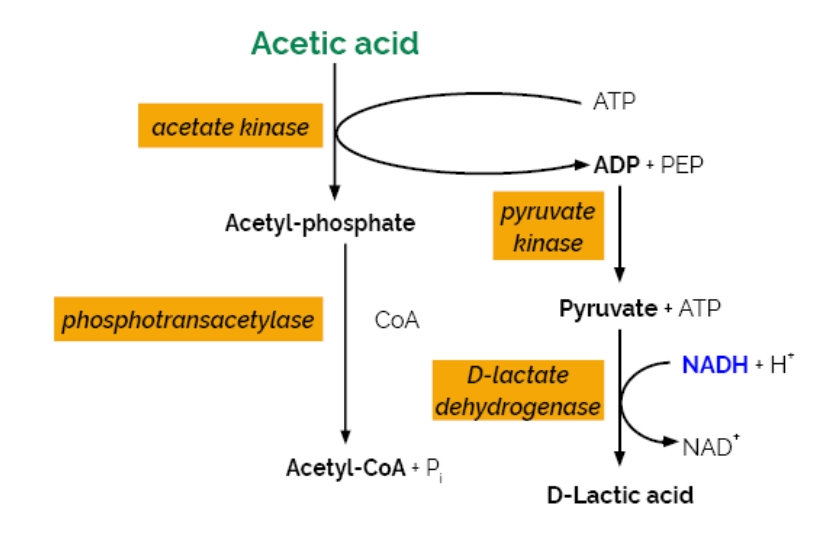
- A. MANUAL ASSAY PROCEDURE:


- B. MICROPLATE ASSAY PROCEDURE:

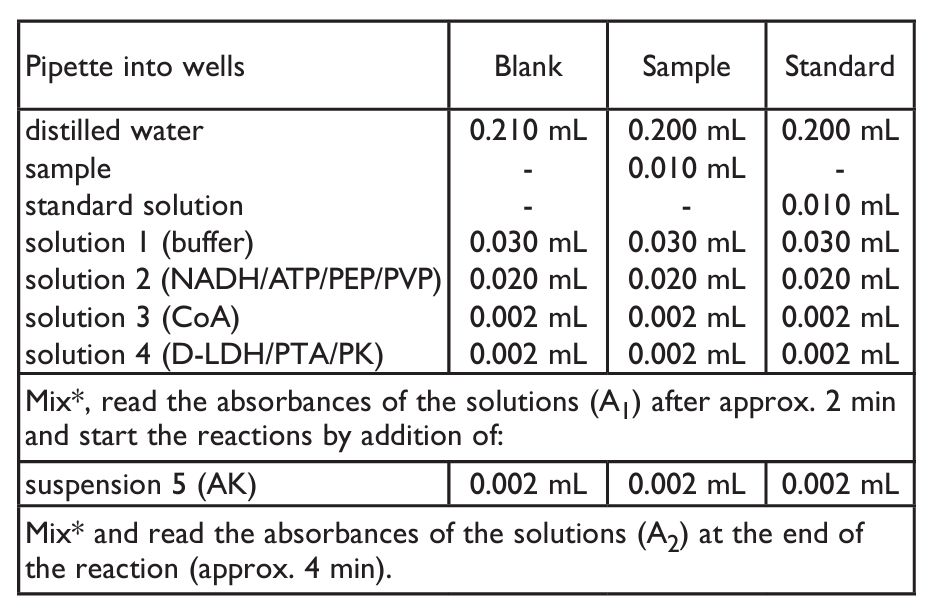

- SAMPLE PREPARATION:
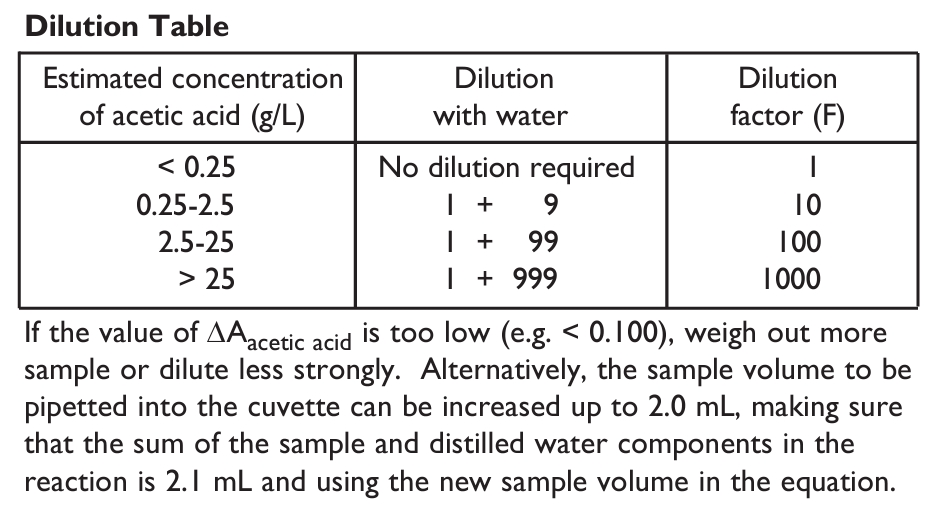
1. Sample dilution.
The amount of acetic acid present in the cuvette (i.e. in the 0.1 mL of sample being analysed) should range between 0.3 and 25 μg. The sample solution must therefore be diluted sufficiently to yield an acetic acid concentration between 0.0003 and 0.25 g/L.
2. Sample handling.
Acetic acid is volatile, so care should be taken when drying or otherwise heating samples containing this analyte in the acid form. Problems associated with the volatility of acetic acid can be minimised by converting it to the salt form (e.g. sodium acetate or potassium acetate). This is achieved by adjusting the pH of the sample to approx. 7.5 using 1 M NaOH or KOH before drying or heating at elevated temperatures.
3. Sample clarification.
a. Solutions:
Carrez I solution. Dissolve 3.60 g of potassium hexacyanoferrate (II) {K4[Fe(CN)6].3H2O} (Sigma cat. no. P9387) in 100 mL of distilled water. Store at room temperature.
Carrez II solution. Dissolve 7.20 g of zinc sulphate (ZnSO4.7H2O) (Sigma cat. no. Z4750) in 100 mL of distilled water. Store at room temperature.
Sodium hydroxide (NaOH, 100 mM). Dissolve 4 g of NaOH in 1 L of distilled water. Store at room temperature.
b. Procedure:
Pipette the liquid sample into a 100 mL volumetric flask which contains approx. 60 mL of distilled water, or weigh sufficient quantity of the sample into a 100 mL volumetric flask and add 60 mL of distilled water. Carefully add 5 mL of Carrez I solution, 5 mL of Carrez II solution and 10 mL of NaOH solution (100 mM). Mix after each addition. Fill the volumetric flask to the mark, mix and filter.
4. General considerations.
(a) Liquid samples: clear, slightly coloured and approximately neutral, liquid samples can be used directly in the assay.
(b) Acidic samples: if > 0.1 mL of an acidic sample is to be analysed undiluted (such as wine or fruit juice), the pH of the solution should be increased to approx. 7.4 using 2 M NaOH, and the solution incubated at room temperature for 30 min.
(c) Carbon dioxide: samples containing a significant amount of carbon dioxide, such as beer, should be degassed by increasing the pH to approx. 7.4 with 2 M NaOH and gentle stirring, or by stirring with a glass rod.
(d) Coloured samples: an additional sample blank, i.e. sample with no AK, may be necessary in the case of coloured samples.
(e) Strongly coloured samples: if used undiluted, strongly coloured samples should be treated by the addition of 0.2 g of polyvinylpolypyrrolidone (PVPP)/10 mL of sample. Shake the tube vigorously for 5 min and then filter through Whatman No. 1 filter paper.
(f) Solid samples: homogenise or crush solid samples in distilled water and filter if necessary.
(g) Samples containing fat: extract such samples with hot water at a temperature above the melting point of the fat, e.g. in a 100 mL volumetric flask. Adjust to room temperature and fill the volumetric flask to the mark with water. Store on ice or in a refrigerator for 15-30 min and then filter. Discard the first few mL of filtrate and use the clear supernatant (which may be slightly opalescent) for assay. Alternatively, clarify with Carrez reagents.
(h) Samples containing protein: deproteinise samples containing protein with Carrez reagents.
商品特色
商品規格
- 商品規格
72 assays (manual) / 720 assays (microplate)
Bottle 1:
Buffer (24 mL, pH 7.4) and sodium azide (0.02% w/v) as a preservative.
Stable for > 2 years at 4°C.
Bottle 2:
NADH, ATP, PEP and PVP. Lyophilised powder.
Stable for > 2 years below -10°C.
Bottle 3: *2
CoA, lyophilised powder.
Stable for > 2 years below -10°C.
Bottle 4:
D-Lactate dehydrogenase, phosphotransacetylase and pyruvate kinase suspension (1.5 mL).
Stable for > 2 years at 4°C.
Bottle 5:
Acetate kinase suspension (1.5 mL).
Stable for > 2 years at 4°C.
Bottle 6:
Acetic acid standard solution (5 mL, 0.10 mg/mL).
Stable for > 2 years; store sealed at 4°C.